Water Balance
Water balance is the interaction of pH, pH buffer (total alkalinity) and water hardness. Fortunately, New Zealand water is naturally clean, fairly soft and has a consistent pH. It usually presents few problems in achieving correct water balance.
It is essential that pool water is kept correctly balanced otherwise corrosion or scale formation may occur on pool fittings, piping, etc, sanitisers will not work properly, and the water will neither feel nor look pleasant.
pH
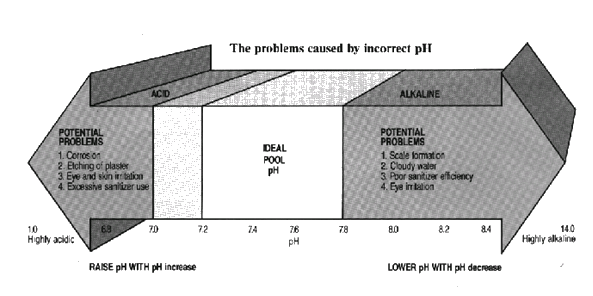
This is a measure of acidity or alkalinity of water. It is measured on a scale ranging from 1 to 14.
A pH of 7.0 is neutral
Above pH 7 the water is alkaline
Below pH 7 the water is acidic.
Pool pH should be maintained between 7.2 and 7.8.
Water with low pH can cause eye and skin irritation, corrosion, etching of plaster and excessive use of sanitiser. Bluewater pH increase is used to correct it. (See page 37).
Water with high pH can result in scale formation, cloudy water, eye irritation and poor sanitiser efficiency. Bluewater pH decrease is used to correct it. (See page 36).
pH is affected by the type of pool surface, heavy rain, wind, dust, evaporation, bather load and the kind of sanitiser or other chemicals used.
pH level should be tested weekly, using the appropriate test kit.
pH Buffer (Alkalinity)
This is often called total alkalinity. It refers to the amount of carbonates, bicarbonates and hydroxides dissolved in pool water.
pH buffer is measured in parts per million (p.p.m.).
For Baquacil sanitised pools the ideal range is between 80 and 150 p.p.m.
For pools sanitised with Bluewater Granular Pool Chlorine, EconoChlor, Stabilised Pool Chlorine, Liquid Pool Chlorine or Salt, the ideal range is 120-150 p.p.m.
For pools sanitised with Bluewater Stabilised Pool Chlorine Tablets the ideal range is 150-200 p.p.m. The reason for this higher level is because the tablets are highly acidic and the buffer level is needed to maintain a correct pH.
The importance of the level of pH buffer is that it acts as a buffer to change in pH. That is, it reduces the reaction to factors that affect water pH. For example, heavy rain can cause the pool to become acidic and lower the pH; the buffer reacts with some of the acid and so reduces the effect.
If the pH buffer level is too low, the pH of the pool will be susceptible to rapid change. This may result in any of the problems associated with incorrect pH.
A high level of pH buffer is not so serious but it will mean that pH adjustment requires more chemicals. The most effective way a high level of pH buffer can be adjusted is by dilution with additional mains or tap water.
Bluewater pH Buffer is used to adjust the buffer level. (See pages 35-36.)
pH buffer level should be tested at the start of each season and monitored each month during the time the pool is in use.
When anything acid or alkaline is added to pool water low in pH buffer, the pool water pH will alter accordingly.
The following example shows how rainwater (slightly acidic) lowers the pH of the pool.
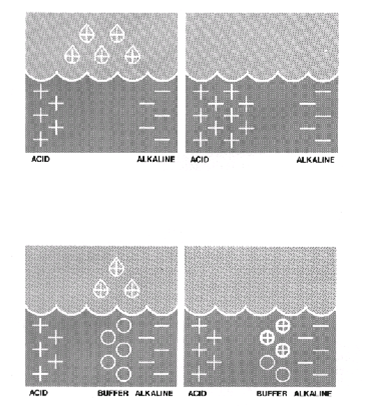
Where the correct level of pH buffer is present, it helps neutralise the acid effect of the rainwater.
Water Hardness
Hardness refers to the amount of calcium and magnesium salts in the pool water. In fact, only calcium harness is important for pool water.
Calcium hardness is measured in parts per million (p.p.m.). The level should be between 100 and 300 p.p.m.
New Zealand water is usually naturally low in calcium. It needs adjusting because low calcium hardness can cause etching of plaster and corrosion.
Calcium hardness is increased by adding Bluewater Water Hardener.
(See pages 38-39).
Lowering calcium hardness is not so easy, but high levels are not usually a problem in New Zealand because top-up water has a low level, so the pool hardness is constantly diluted.
Water hardness should be checked at the beginning and end of each swimming season.
Achieving Water Balance
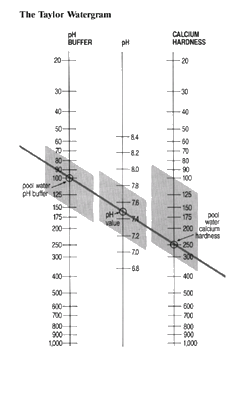
Calcium hardness, pH and pH buffer levels must be in balance if pool water is to look right, feel right, and be free of problems.
The simplest way of checking the balance is to use the Taylor Watergram.
Check the levels in the pool of pH, pH buffer, and water hardness. On the Watergram draw an imaginary line between pH buffer and hardness to find the pH at which the pool water is balanced.
Remember that the pH must be in the range 7.2 to 7.8, because outside this range eye and skin irritation may occur.
The line through the three measurements must be straight, but it need not slope in the direction shown. For example a pool with a pH buffer of 150 p.p.m. and calcium hardness of 125 p.p.m. would be balanced with a pH of 7.6. The range of water balance parameters demonstrates there is considerable latitude in the levels that can give a satisfactory balance.
Total Dissolved Solids
Total dissolved solids is a measure of material in solution that is not removed by filtration or chemicals. It can affect water balance but is rarely a problem because it is overcome by dilution. Most pools are regularly diluted by being topped up to replace water lost through evaporation and splashing. Total dissolved solids, therefore, rarely build up to problem proportions.
